 However, to fill the cost gap, considerable efforts will be required. These electrolyzers are intended for use in transportation (HGV stations and large fleets) and industry to compete with steam forming. Improvements in the reactivity of these pressure-increasing electrolyzers have recently been achieved.Ĭurrently, using mature bipolar technologies, the effort is focused on heavy modular architectures that could reach 100 MW (25 × 4 MW) capable of producing more than 40 tons per day of H 2 (and eight times more oxygen). Its good performance (60%–70%) is associated with high inertia that makes it unsuitable for rapid fluctuations in intermittent electrical sources. Power consumed: 4.5–5 kWh/N m 3 of hydrogen producedĪlkaline electrolysis works at an average temperature (80–160☌) and a moderate pressure (3–30 bars) with potassium as a liquid electrolyte. Read moreĭiaphragm: asbestos, sintered nickel or microperforated canvas This chapter addresses the learning rate of alkaline electrolysis systems from the period 1956–2016, issues with generating experience curves for the system as a whole as opposed to on a component basis, associated cost reduction drivers, and the future outlook of the alkaline electrolysis market until 2030. But in recent years, plant scale has increased (10 MW with few at 100 MW) compared to scale in the 1990s in response to increased demand for green hydrogen and moving away from hydrogen production from fossil fuels. With the emergence of cheap hydrogen from steam methane reforming from the late 1980s, the production of small-scale plants (around 1 MW) dominated the electrolysis market. Gert Jan Kramer, in Technological Learning in the Transition to a Low-Carbon Energy System, 2020 AbstractĪlkaline electrolysis as an industrial process has been around since the advent of commercial power at the beginning of the 20th century with most large-scale plants (up to 165 MW) built between the 1920s and 1980s in response to hydrogen demand for the ammonia industry. Therefore, current studies on AEC mainly focus on enhancing electrochemical performance, pressurizing, and dynamic operation for addressing intermittent renewable energy. The electrical efficiency of AEC is 62%–82%, and an AEC system generally requires 4.5–6.6 kWh electricity per Nm 3 H 2 production. This means that electricity consumption of AEC is ∼19% higher than that of PEMFC, and 46% higher than that of SOEC for the same hydrogen production. AEC generally operates at 1.8–2.4 V, higher than PEMEC and SOEC. 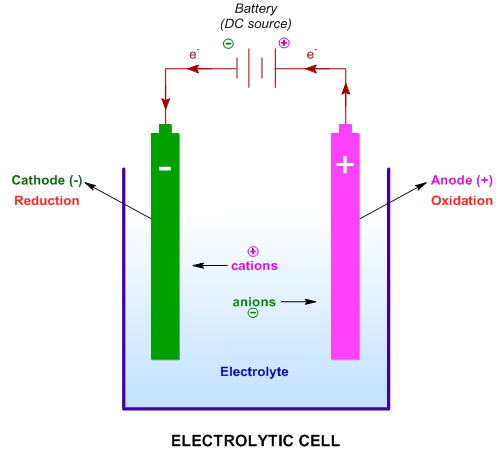
Slower electrochemical reaction rate of AEC indicates that larger effective reaction area (larger volume) is needed than PEMEC and SOEC for the same hydrogen production rate. 
Currently, the maximum operable current density of AEC is around 400 mA cm −2, only 20% of that of PEMEC or SOEC. Fig. 3.6 shows the comparison among three types of electrolyzers. However, AEC has similar issues to AFC, such as the corrosion and leakage of aqueous electrolyte, slow electrochemical kinetics and high polarization losses (high electricity consumption). The stack lifetime of AEC can reach as long as 60,000–90,000 h (7–10 years). 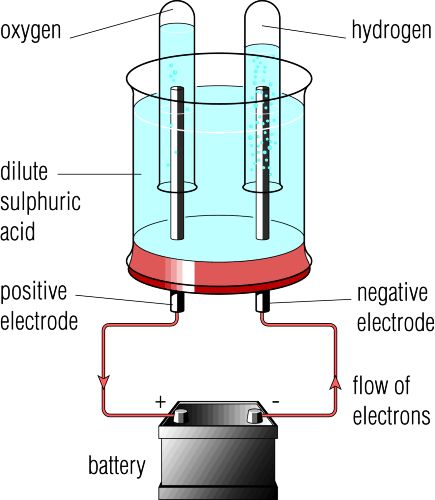
Currently, AEC is the most developed electrolyzer and also has the lowest capital cost (1000–5000 $/kW varying with scale ). The electrolyte of AEC is 25–30 wt.% KOH or NaOH solution. AEC, the reversed process of AFC, is the first proposed electrolyzer among all the electrolyzers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
